UCLA Scientists Uncover Senescent Macrophages as a Key Driver of Liver Aging and Fatty Liver Disease
UCLA scientists have identified a previously underestimated group of immune cells, known as senescent macrophages, that accumulate in aging tissues and are particularly prevalent in the livers of individuals with fatty liver disease. This groundbreaking discovery, published in the prestigious journal Nature Aging, reveals that the targeted removal of these "zombie cells" in mice led to a significant reduction in inflammation and a reversal of liver damage, even when the animals maintained an unhealthy diet. The findings offer a promising new avenue for understanding and potentially treating age-related diseases and metabolic disorders.
Cellular senescence, a state where cells cease to divide but remain metabolically active and release pro-inflammatory molecules, has long been recognized as a contributor to aging and various diseases. However, precisely identifying and characterizing senescent cells, especially within the complex immune system, has been a significant challenge. The UCLA team’s work zeroes in on macrophages, critical immune cells responsible for clearing cellular debris and pathogens, and elucidates their role in this detrimental process.
The Macrophage Enigma: Identifying the Senescent Signature
For years, the scientific community debated whether macrophages, which already exhibit certain molecular characteristics similar to senescent cells in their healthy state, could indeed become senescent themselves. This ambiguity made it difficult to distinguish between normally functioning macrophages and those contributing to pathological inflammation.
"Senescent cells are fairly rare, but think of them like a broken-down car on the 405," explained Anthony Covarrubias, senior author of the study and a member of the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA. "Just one stalled car can back up traffic for miles. Now imagine five or ten of them slowly accumulating. That’s what these cells do to a tissue: even a small number causes enormous disruption."
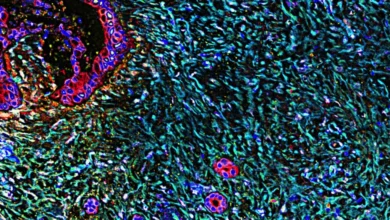
The UCLA researchers overcame this challenge by pinpointing a distinct molecular signature. They discovered that the co-expression of two specific proteins, p21 and TREM2, reliably identifies macrophages that have entered a senescent state, are no longer performing their intended cleanup functions, and are actively promoting inflammation in surrounding tissues.
This newly identified marker allowed for a quantitative assessment of senescent macrophage accumulation. In young mice, senescent macrophages constituted a mere 5% of the liver macrophage population. In contrast, older mice exhibited a dramatic increase, with senescent macrophages making up between 60% and 80% of the total. This surge directly correlated with the observed increase in chronic liver inflammation associated with aging.
Cholesterol: A Critical Catalyst for Macrophage Senescence
Beyond the natural aging process, the study uncovered a significant trigger for the development of senescent macrophages: excess cholesterol. In laboratory experiments, exposing healthy macrophages to high levels of low-density lipoprotein (LDL) cholesterol induced a senescent phenotype. These cells stopped dividing, began secreting inflammatory proteins, and displayed the characteristic p21-TREM2 signature.
"Physiologically, macrophages can handle cholesterol metabolism," stated Ivan Salladay-Perez, the first author of the study and a graduate student in the Covarrubias lab. "But in a chronic state, it’s pathological. And when you look at fatty liver disease, which is driven by overnutrition and too much cholesterol in the blood, that excess cholesterol appears to be a major driver of the senescent macrophage population."
This finding has profound implications, suggesting that diets rich in fat and cholesterol may accelerate biological aging by promoting macrophage senescence not only in the liver but potentially in other vital organs, including the brain, heart, and adipose tissue. This broadens the scope of potential age-related diseases linked to this cellular dysfunction.
Reversing Liver Damage: The Power of Senescent Cell Clearance
To ascertain whether the removal of these detrimental cells could translate into tangible health benefits, the researchers treated mice with ABT-263, a drug specifically designed to eliminate senescent cells. The results were nothing short of remarkable.
Mice that had been fed a high-fat, high-cholesterol diet, exhibiting significant liver enlargement and weight gain, showed dramatic improvements after treatment. Their liver size decreased from approximately 7% of body weight to a healthier range of 4-5%. Concurrently, their body weight dropped by about 25%, from roughly 40 grams to around 30 grams.
Visually, the treated livers were notably smaller and appeared healthier, with a normal red hue, starkly contrasting with the enlarged, yellowish livers of untreated control animals.
The study’s authors were particularly struck by the fact that eliminating senescent macrophages alone led to substantial metabolic improvements, even in the absence of dietary changes. "That’s what wowed me," Salladay-Perez remarked. "Eliminating senescent cells doesn’t just slow the fatty liver — it actually reverses it." This suggests that senescent macrophages play a pivotal and reversible role in the pathogenesis of fatty liver disease.
Human Relevance: Evidence in Liver Biopsies
The researchers then sought to determine if their findings in mice extended to human health. By analyzing an existing genomic dataset from human liver biopsies, they discovered that the same senescent macrophage signature (p21-TREM2) was significantly elevated in diseased livers compared to healthy ones. This strongly suggests that macrophage senescence is a contributing factor to chronic liver disease in humans.
The implications of this discovery are particularly urgent in regions like Los Angeles, where an estimated 30-40% of the population suffers from fatty liver disease, with even higher prevalence rates observed in Latino communities. Current treatment options are limited, and effective early detection tools remain elusive, underscoring the critical need for novel therapeutic strategies.
"This is a huge public health crisis in the making," emphasized Covarrubias, who also holds a position as an assistant professor of microbiology, immunology, and molecular genetics. "We’re seeing fatty liver disease in younger and younger people. So we’re really happy to make some inroads into understanding what’s driving it and identifying cell types we might be able to target."
Future Directions: Towards Safer Therapies and Broader Applications
While ABT-263 proved effective in mice, its toxicity profile makes it unsuitable for widespread human use. The UCLA team is now focused on identifying and screening for safer compounds that can selectively eliminate senescent macrophages without eliciting harmful side effects. This pursuit of targeted senolytics, drugs that clear senescent cells, represents a significant frontier in regenerative medicine.
Furthermore, the researchers are actively investigating whether similar mechanisms of senescence are at play in other age-related diseases. For instance, in the brain, microglia, the resident macrophages of the central nervous system, may undergo senescence when exposed to the accumulated cellular debris characteristic of neurodegenerative conditions like Alzheimer’s disease. This line of inquiry could unlock new therapeutic targets for a range of debilitating neurological disorders.
A Unifying Mechanism: Aging and Disease Intertwined
The UCLA study’s findings provide robust support for the geroscience hypothesis, which posits that a common underlying process of aging drives multiple age-related diseases. In this context, the accumulation of senescent macrophages could serve as a central hub, contributing to conditions as diverse as fatty liver disease, atherosclerosis, Alzheimer’s disease, and even certain types of cancer.
"If you really understand the basic mechanisms driving inflammation with aging, you can target those same mechanisms to treat not just fatty liver disease, but atherosclerosis, Alzheimer’s and cancer," Salladay-Perez articulated. "It all goes back to understanding how these cells arise in the first place."
This research signifies a pivotal step forward in unraveling the intricate relationship between aging, inflammation, and disease. By identifying and characterizing senescent macrophages, the UCLA team has opened a critical new avenue for developing interventions that could not only treat specific diseases but also promote healthier aging overall.
The research was made possible through crucial funding from the National Institutes of Health, the Glenn Foundation for Medical Research, the American Federation for Aging Research, and the UCLA-UCSD Diabetes Research Center, highlighting the collaborative and multi-faceted nature of scientific advancement. The continued exploration of senolytic therapies holds immense promise for improving human healthspan and mitigating the burden of age-related chronic diseases.