Brazilian Researchers Engineer Supercharged Natural Killer Cells for Enhanced Cancer Immunotherapy
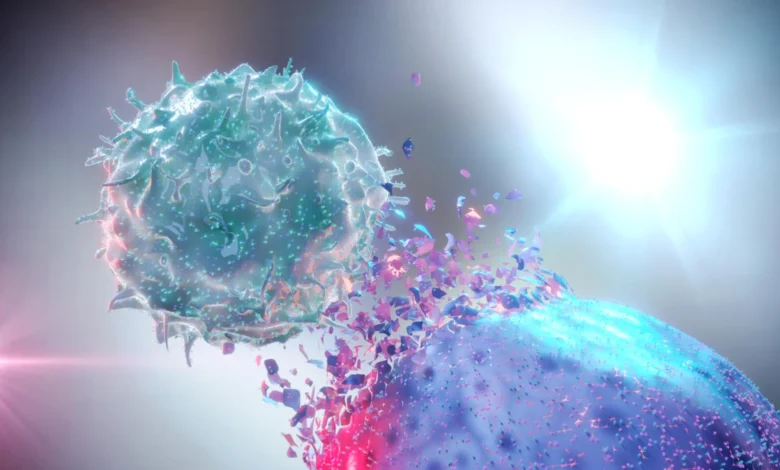
Researchers in Brazil have achieved a significant breakthrough in the field of cancer immunotherapy, developing a novel approach to augment the power and precision of natural killer (NK) cells. This innovative strategy, detailed in a recent study published in Frontiers in Immunology, focuses on equipping NK cells with enhanced signaling capabilities and introducing a controlled method for their activation, potentially paving the way for more effective and adaptable cell-based cancer treatments.
The study, conducted by scientists at the Ribeirão Preto Blood Center and the Center for Cell-Based Therapy (CTC), utilized the well-established NK-92 cell line as a platform to engineer advanced chimeric antigen receptors (CARs). CARs are synthetic receptors designed to direct immune cells, such as NK cells, to specifically recognize and attack cancer cells. The Brazilian research team’s innovation lies in the intricate design of these CARs, incorporating specific costimulatory components known to amplify NK cell activation signals. Among these crucial additions are the 2B4 and DAP12 signaling pathways.
The Science Behind Supercharged NK Cells: Unlocking Potent Anti-Tumor Activity
Natural killer cells are a vital component of the innate immune system, playing a critical role in identifying and eliminating cells that are infected or cancerous without prior sensitization. Their inherent ability to detect and destroy abnormal cells makes them an attractive candidate for cancer immunotherapy. However, to harness their full therapeutic potential, particularly in the context of CAR-based strategies, researchers must overcome challenges related to their activation and sustained efficacy.
CAR-based therapies have already revolutionized the treatment landscape, demonstrating remarkable success, especially in combating hematological malignancies like certain types of leukemia and lymphoma. While CAR-T cell therapy, which engineers T lymphocytes, has garnered considerable attention and clinical validation, the development of equally potent and reliable CAR-NK cell therapies is an ongoing area of intense research. A central question in this field is how to optimize the internal signaling mechanisms within NK cells to ensure they are primed for optimal anti-tumor activity.
The groundbreaking research from Brazil directly addresses this critical knowledge gap. By meticulously investigating the influence of specific internal signaling domains on NK cell function, the scientists were able to identify key pathways that dramatically enhance cellular responsiveness. The incorporation of the 2B4 and DAP12 signaling moieties into the CAR construct proved to be a pivotal step. These components are integral to the natural activation cascades of NK cells, and their integration into the engineered CAR effectively primes the NK-92 cells, making them "ready to attack" cancer cells with significantly improved efficacy. This enhanced activation state translates to a more robust and potent destruction of tumor cells in preclinical settings.
A Dual Approach: Enhancing Activation and Introducing Precise Control
Beyond simply boosting the intrinsic activating signals of NK cells, the Brazilian research team also explored a sophisticated strategy to exert finer control over their therapeutic activity. This involved investigating the potential of a temporary, drug-based modulation of cell function. The scientists tested the effects of dasatinib, a well-known tyrosine kinase inhibitor, which can transiently suppress cellular activity. The rationale behind this approach was to explore whether controlled "pauses" in NK cell function, induced by a specific drug, could ultimately lead to improved overall performance and safety.
The experimental findings from this dual approach were highly encouraging. The integration of optimized activation signals, such as those provided by 2B4 and DAP12, in conjunction with the reversible pharmacological control offered by dasatinib, demonstrated a synergistic effect. This combination not only amplified the intrinsic strength of the CAR-NK cells but also enhanced their efficiency in targeting and eliminating tumor cells. This sophisticated strategy holds immense promise for the future design of cell-based cancer therapies that are not only potent but also possess a greater degree of controllability, a crucial factor for therapeutic safety and patient well-being.
Preclinical Validation: Superior Tumor Control in Animal Models
The efficacy of these engineered CAR-NK cells was rigorously tested in preclinical models. According to information released by the Ribeirão Preto Blood Center Press Office, experiments conducted with animal models yielded highly promising results. Specifically, CAR-NK cells that were engineered with the enhanced 2B4-DAP12 signaling pathway and subsequently pretreated with dasatinib exhibited a superior ability to control tumor growth compared to CAR-NK cells employing more conventional designs. This preclinical evidence provides a strong foundation for the translation of these findings into potential clinical applications, suggesting that this novel approach could lead to more effective management of cancer in living organisms. The ability to achieve better tumor control in these models is a significant indicator of the therapeutic potential of this advanced immunotherapy strategy.
A Collaborative Endeavor: Fostering Innovation through Institutional Support
This pioneering research is a testament to the power of collaboration and robust institutional support for scientific advancement. The Center for Cell-Based Therapy (CTC) is a key initiative within the Research, Innovation, and Dissemination Centers (RIDCs) framework, a program generously supported by the São Paulo Research Foundation (FAPESP). The CTC operates under the umbrella of the Ribeirão Preto Blood Center and is closely affiliated with the Hospital das Clínicas, the general and teaching hospital associated with the Ribeirã Preto Medical School of the University of São Paulo (FMRP-USP). This integrated structure fosters a dynamic environment where cutting-edge research can be translated into tangible improvements in healthcare.
The Ribeirão Preto Blood Center, a leading institution in blood research and transfusion medicine, provides a critical infrastructure and expertise for cell therapy development. Its affiliation with FMRP-USP ensures access to a rich academic environment, facilitating interdisciplinary collaboration and the training of future scientists. The support from FAPESP is instrumental in funding these ambitious research projects, enabling scientists to explore novel therapeutic avenues and push the boundaries of medical science.
Broader Implications: A New Era of Controllable Cancer Therapies
The implications of this research extend far beyond the immediate findings. The successful demonstration of enhanced NK cell activation through specific costimulatory pathways, coupled with the innovative use of pharmacological control, signals a potential paradigm shift in the development of cell-based cancer immunotherapies.
For patients, this could translate to more effective treatments with potentially fewer side effects. The ability to fine-tune the activity of CAR-NK cells offers a pathway to greater precision in targeting cancer cells while minimizing damage to healthy tissues. This enhanced control is particularly crucial for managing potential on-target, off-tumor toxicities, a concern in many CAR-based therapies.
Furthermore, the research contributes to a deeper understanding of NK cell biology and signaling. By dissecting the intricate mechanisms that govern NK cell activation, scientists are building a more comprehensive knowledge base that can inform the design of future immunotherapies. The insights gained from this study could be applied to a broader range of cancers and immune cell types, accelerating progress across the entire field of immunotherapy.
The development of CAR-NK cells with built-in safety switches or controllable activation mechanisms is a significant step towards personalized medicine. The ability to tailor the therapy to individual patient needs and tumor characteristics could lead to improved treatment outcomes and a more efficient use of healthcare resources.
Looking Ahead: From Bench to Bedside
While the results are highly promising, it is important to acknowledge that this research is still in its preclinical stages. The journey from laboratory discoveries to approved clinical treatments is often long and complex, involving rigorous testing in human clinical trials to ensure both safety and efficacy. However, the scientific rigor and innovative nature of this Brazilian study provide a strong foundation for optimism.
The findings published in Frontiers in Immunology represent a significant advancement in the quest for more potent, precise, and controllable cancer immunotherapies. The ongoing work by researchers at the Ribeirão Preto Blood Center and the CTC, supported by institutions like FAPESP, is crucial for unlocking the full potential of natural killer cells and bringing us closer to a future where cancer is a more manageable and treatable disease. This research highlights Brazil’s growing contribution to global biomedical innovation and its commitment to developing cutting-edge solutions for pressing health challenges. The next steps will likely involve further optimization of the CAR designs, expanded preclinical testing, and eventually, progression into early-phase human clinical trials. The scientific community will be keenly watching as this promising avenue of research unfolds.